ARTÍCULO |
Curcumin induces apoptosis of 3T3-L1 adipocytes and affects molecular signals of adiponectin, AMPK and PKA
Ebtesam A. Al- Suhaimi
Biology Department, College of Sciences, University of Dammam, Dammam, Saudi Arabia.
*e-mail: ealsuhaimi@ud.edu.sa
An. Real Acad. Farm. Vol. 80, Nº 4 (2014), pag. 720-734
abstract
Apoptosis is an important cellular physiological function to continually balance the cell proliferation with cell death to maintain the healthy internal milieu. This study assessed the ability of Curcumin to inhibit 3T3-L1 adipocytes viability and its effects on apoptosis to identify a potential molecular approach for preventing adipocytes growth. To optimize the growth inhibitory concentration, 3T3-L1 adipocytes treated with different concentrations of Curcumin (5, 10, 20, 40, 60, and 80 μM) for 24 hours, and were analyzed by MTT and found 40 μM as an optimal dose. Cell cycle was analysed by FACS using the optimal dose for different periods (4-24h). Western blot was carried out using antibodies to detect protein expression and Phosphorylation of Adiponectin, AMPK, CREB, PKA catalytic subunit, and Akt. PKA relative activity and ROS generation was also assessed by DCFHDA fluorescence. The findings of the study revealed that Curcumin has successfully arrested 3T3L1 adipocytes cycle and exerted apoptotic action on the cells in a concentration- time dependent manner; increased Adiponectin expression; activated phosphorylated AMPK (p-AMPK) and inhibited PKA activation. Curcumin prevented the production of ROS generation. Results indicate that Curcumin exerts a very potent apoptotic action on 3T3L1 adipocytes and modulates adiponectin expression and AMPK and PKA signalling. These findings lead us to further investigate and support the potential use of Curcumin therapy in obesity resistance. |
Keywords: Apoptosis; 3T3L1; Curcumin; Adiponectin; AMPK; PKA.
resumen
La curcumina inhibe la viabilidad celular e induce la apoptosis mediante la modulación de la adiponectina, la AMPK y PKA en adipocitos 3T3-L1
La apoptosis es una función fisiológica celular importante para equilibrar de manera continua la proliferación y la muerte celulares con el objeto de mantener el entorno interno sano. Este estudio se determina la capacidad de la curcumina de inhibir la viabilidad de los adipocitos 3T3-L1 y sus efectos sobre la apoptosis para identificar un posible acercamiento molecular para prevenir el crecimiento de los adipocitos. Para optimizar la concentración inhibitoria del crecimiento, los adipocitos 3T3-L1 han sido tratados con diversas concentraciones de curcumina (5, el μM 10, 20, 40, 60, y 80) durante 24 horas y analizados por MTT, encontrando el μM 40 como dosis óptima. El ciclo de la célula ha sido analizado por FACS usando la dosis óptima en diversos períodos (4-24h). El Western blot se realizó usando los anticuerpos para detectar la proteína y la fosforilación de la adiponectina, de la subunidad catalítica de AMPK, de CREB, de PKA, y de Akt. La actividad relativa de PKA y la generación del ROS también fueron determinadas por fluorescencia de DCFHDA. Los resultados del estudio revelaron que la curcumina ha detenido con éxito el ciclo de los adipocitos 3T3L1 y que la acción apoptótica ejercida en las células en una concentración mide el tiempo de manera dependiente; expresión creciente de adiponectina; AMPK fosforilado activado (p-AMPK) y activación inhibida de PKA. La curcumina previno la producción de la generación del ROS. Los resultados indican que la curcumina ejerce una acción apoptótica muy potente en los adipocitos 3T3L1 y modula la expresión de la adiponectina indicando AMPK y PKA. Estos resultados nos conducen a ir más lejos en la investigación y el uso potencial de la terapia de la curcumina para combatir la obesidad. |
Palabras clave: Apoptosis; 3T3L1; curcumina; adiponectina; AMPK; PKA.
1. INTRODUCTION
The possibility of managing cell survival and death has great therapeutic potential given the importance of cell cycle transition and apoptosis in the physiological functions of cells (1). The inhibition of differentiation, adipogenesis, angiogenesis and stimulation of physiological apoptosis in adipocytes in vitro or in vivo may be a potential strategy to inhibit adipose cell expansion and growth, thus preventing obesity and associated disorders. It has recently been discovered that adipose tissue is a hormone-secreting organ that releases adipokines, such as leptin, adiponectin, and visfatin. As such, adipose tissue is a principal of both the endocrine and immune systems (2). 3T3L1 cells are considered to be an excellent model to study adiposity because Adiponectin is primarily distributed in microvesicle particles of adipocytes (3). Many factors include curcumin could modulate leptin release from 3T3L1cells (4, 5). Different mechanisms for physiological and pathological apoptosis processes have been investigated, including suppressing signal transduction pathways to stimulate apoptosis (6). Cyclic adenosine monophosphate (cAMP)-dependent protein kinase (PKA) functions as both a suppressor and activator of apoptosis. In arterial smooth muscle cells adenosine-mediating apoptotic pathways are activated (7); however, Zhang et al, (8) reported that the cAMP signalling pathway has been found to either support or inhibit apoptosis, depending on cellular situation. The adipokine leptin inhibits growth via apoptosis caused by adenylate cyclase/cAMP/PKA-elevating agents in some cancer cells (9). Increase in weight is resulted due to the growth and enlargement of adipose tissue, and AMPK is a major controller of energy metabolism that increases fatty acid oxidation and suppresses lipid accumulation (10,11). Mihaylova and Shaw (12) reported that in addition to physiological AMP/ADP elevation stresses, AMPK can be stimulated by many pharmacological agents and factors, including leptin, and AMPK in turn can suppress cell growth by acting as a metabolic check point. AMPK also directly phosphorylates some regulatory associated proteins and reduces expression of cAMP response element-binding protein (CREB) targets, which is important since CREB activation stimulates the expression of multiple transcription factors required for adipogenesis (13). Reactive oxygen species (ROS) and mitochondria play a prominent role in apoptosis as described by Simon et al. (14). Recently, it was found that apoptosis is regulated by ROS through a variety mechanisms depending on cell and conditioned media types (15). Adipose tissue growth is sensitive to angiogenesis and adipogenesis inhibitors (16), as well as adipokines (2) and these may serve as effective tools for controlling adipocytes growth.
Curcumin was reported as a potent inducer for apoptosis characteristics, including cell fading, chromatin retraction, DNA fragmentation, and cell membrane blebbing (6). The anti-angiogenic effect of curcumin in vivo was supported by its action at the level of gene expression in modified media of cells treated with different doses of curcumin (1 μM–1 mM) for various time durations (0–24 h) (17) . Curcumin has been studied for its potential effect in antiangiogenic activity in tumor (18) and in preventing obesity in C57BL mice and inhibiting adipogenesis in 3T3-L1 adipocytes (16) as well as cancers (19- 21). In contrast, Kim et al (22) found that low concentration of curcumin activated proliferation, improved stemness and migration of 3T3-L1 preadipocytes. Cellular survival required Protein Kinase B (PKB or Akt1) through its inhibiting effect on apoptosis so it is involved as principal factor in multiple cancers (23). But curcumin was able to induce cell cycle arrest in different phases, including human osteosarcoma cells in successive G(1)/S and G(2)/M phases (19) and on colorectal carcinoma cells in the S phase (24). Recent studies described an inhibiting growth action of curcumin by stimulation of adenosine 5'-monophosphate (AMP, also called activated protein kinase (AMPK)) in adipocytes and cancer cells (19) and cause apoptosis in several cell lines (20, 21). The safety of curcumin was proved previously (17, 25- 27)
The current study was performed to investigate the underlying molecular mechanism of curcumin inhibition of 3T3-L1 adipocytes. The study will aid further molecular signals involved in 3T3L1 adipocytes apoptosis and supports the potential use of Curcumin therapy in obesity resistance.
2. materials and methods
2.1. Cell culturing and treatment.
The 3T3-L1 cells were obtained from American Type Culture Collection (ATCC-CL-173) and maintained at 37°C in a 5% CO2-moistened environment. 3T3-L1 cells were cultured in Dulbecco’s minimum essential medium (DMEM) at a density of 5 × 105 and supplemented with L- glutamine, 10% heat-inactivated fetal bovine serum (FBS), and penicillin-streptomycin (Gibco, Invitrogen Corporation, Carlsbad, CA, USA). After cells reached 70% confluence, Then cells were collected and sub cultured using a standard trypsin /EDTA method (28 - 30 ). Different concentrations of curcumin were dissolved in DMSO for cells treatment as well as with 40 µM for different periods (h).
2.2. Cell viability assay.
MTT ( 3-(4, 5- dimethylthiazol-2-yl)-2, 5- diphenyl tetrasodium bromide) reagent was used for detecting the cell death induce by curcmin treatment according to the previously reported method (31). Cells were treated with different doses of curcumin for 24 h, followed by the addition of MTT reagents and finally DMSO . After discarding unbound stain, the crystalised formazan was dissolved and spectrophotometrically measured at 570 nm. Growth inhibition % was presented, with control growth as 100%, viability% was calculated (sample/control × 100) (32). Cell culturing was repeated to evaluate the dose-dependent effect of curcumin using concentrations of (5, 10, 20, 40, 60, and 80 μM) for 24 h. Ultimately, 40 μM curcumin was used to evaluate the time-dependent effect of treatment.
2.3. Flow cytometric analysis of cell cycle status.
After treatment with (40 µM) curcumin for different time periods (4, 8 ,12, and 24 h), cell cycle progress was examined by fluorescence-activated cell sorting (FACS). In brief, cells were collected by centrifugation. For fixation cells were vortexed slowly with 70% ethanol and kept at 4°C overnight, stained with propidium iodide (PI) reagents, the stained DNA content was then measured using a flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA) as previously described(29).
2.4. Western blotting.
After treatment with 40 µM curcumin, the cells were washed with ice-cold 1× phosphate-buffered saline (PBS), collected, centrifuged, and total protein was extracted using radioimmunoprecipitation buffer. In brief, lysis buffer were add to re-suspend the pellets, which were subjected to ultrasonication, and then centrifuged to separate cell debris. Supernatants were kept at – 20°C until use (9, 33). Immunoblotting was carried out using antibodies against β-actin, PKA (ab5816, abcam, Cambridge, UK), p-CREB (9191s Cell Signaling Technology, Danvers, MA, USA), CREB (9197, Cell Signaling Technology), p-AMPK (2535s, Cell Signaling Technology), and AMPK (2532, Cell Signaling Technology) according to the manufacturers’ protocols.
2.5. Protein Kinase A (PKA) phosphorylation assay.
PKA activity was measured based on transferring 32P-ATP to a targeted peptide substrate. Relative PKA activity was measured with a liquid scintillation counter as previously described (34).
2.6. Measurement of reactive oxygen species (ROS) generation.
Intracellular ROS concentrations were assessed using the oxidant-sensitive fluorescent probe-dichloro-dihydro-fluorescein diacetate (DCFHDA. 3T3L1 cells were incubated with DCFHDA and then washed with 1× PBS. Fluorescence was measured spectrofluorometery at wavelengths (excitation 507 nm and emission: 530 nm) as previously described (35).
2.7. Statistical analysis.
Three independent experiments were carried out for the statistical evaluation. Data are presented as mean ± SEM. All results were generated with SPSS (Chicago, IL, USA) using analysis of variance (ANOVA) and Least Significant Difference (LSD) post-hoc tests. Values of P < 0.05 were considered significant.
3. results
3.1. Curcumin inhibits 3T3L1 adipocyte viability in a dose- and time-dependent manner.
The cell life survival of curcumin (viability or % inhibition) was measured with the MTT cell viability assay post-induction and confirmation of adipocyte differentiation. The results revealed that treatment of 3T3L1 adipocytes for 24 h with different concentrations of curcumin (5, 10, 20, 40, 60, and 80 µM) in the culture media resulted in significant (p<0.01) and gradual inhibition in mean cell viability (94.76% ± 1.01%, 89.65% ± 0.65%, 84.64% ± 0.93%, 71.90% ± 1.09%, 51.51% ± 1.42%, 28.91% ± 6.65%, respectively; vs compared to control cell (viability of 100%; (Figure 1)).
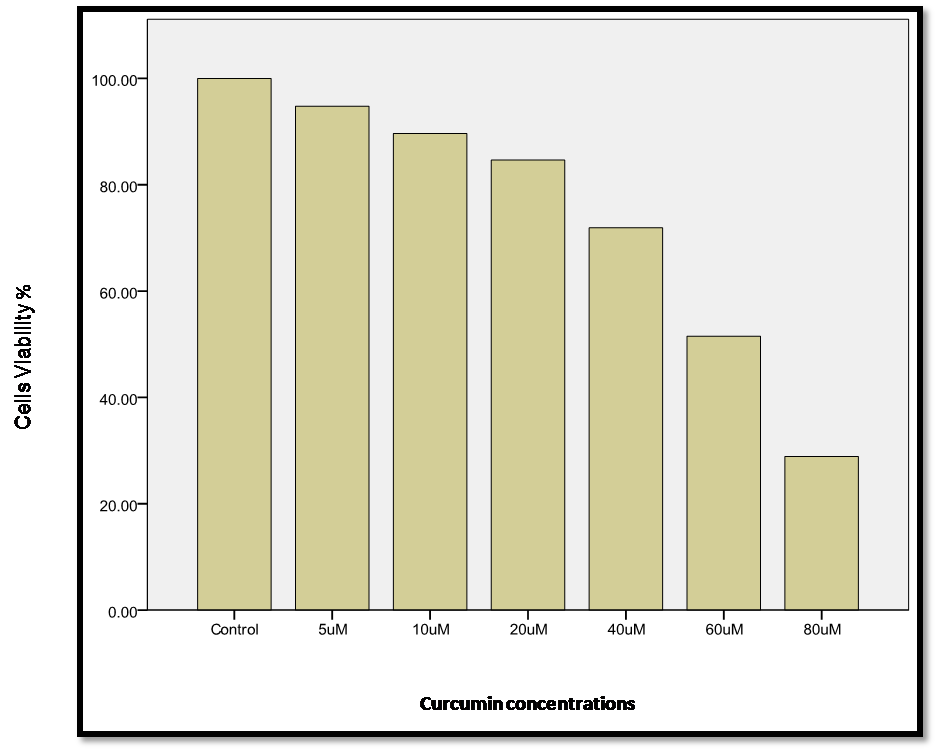
Figure 1.- Viability % inhibition effect of different concentrations of curcumin ( 5- 80 µM ) on 3T3L1 adipocytes. Cells were incubated with curcumin for 24h. Viability of the cells calculated by formula sample/control) x100.
Post-hoc testing (LSD) showed that the most significant inhibition (P ≤ 0.01) started from the dose of 40 µM, which is physiologically relevant. Moreover, induction of maximum inhibition by curcumin - as previously recommended- at (P ≤ 0.01) with this concentration (Figure 1). The selected dose (36- 37) showed a clear gradual inhibition in 3T3L1 cell viability% in a time-dependent manner (Figure 2), indicating that cell viability was significantly decreased (P ≤ 0.01) with longer incubations (97.13% ± 0.85%, 90.42% ± 1.44%, 74.38% ± 1.87%,and 64.12% ± 3.54% at 4, 8, 12, and 24 h, respectively).
3.2. Curcumin induces apoptosis.
FACS analysis provided information about the extent of cell death (%). The results clearly showed that curcumin (40 μM) induced apoptosis in a time-dependent manner, indicated an increase in cell death % compared with control (Figure 3).
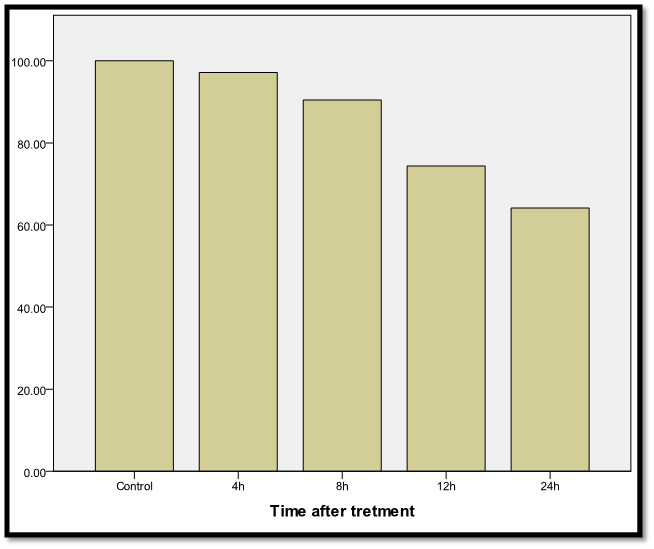
Figure 2.- Viability % inhibition effect of 40 µM curcumin for different periods ranged from (4-24h) on 3T3-L1 adipocytes. Cells Viability % was calculated by formula sample/control) x100.
3.3. Curcumin induces AMPK and adiponectin activation and suppresses PKA and CREB phosphorylation.
We next studied downstream the effect of Curcumin on these signals. Western blot analysis demonstrated that cells treated with 40 μM curcumin induced AMPK phosphorylation (p-AMPK) and reduced levels of phosphorylated CREB (p-CREB) and phosphorylated Akt (p-Akt, to a lesser extent). Curcumin increased the expression of adiponectin, however, total AMPK expression was unaffected. β-actin was used as a loading control (Figure 4).
3.4. Curcumin inhibits PKA activation.
We further demonstrated a gradual decrease with time in the expression of the PKA catalytic subunit, an important transcription factor. The results of the PKA relative phosphorylation assay support this result; PKA relative activity was inhibiting 4–24-h post-curcumin treatment (Figure 5B)

Figure 3.- Curcumin (40 μM) induces apoptosis (the number of cell death (%)) with time increasing.
3.5. Curcumin-inhibits oxidative stress-induced apoptosis.
ROS relative fluorescence was measured after curcumin treatment (40 μM) showed an inhibitory effect on ROS generation level with/without H2O2 treatment of 3T3L1 adipocytes (Figure 6).
4. discusion
This present study was performed to address contrasting data demonstrating that Curcumin inhibits adipogenesis in 3T3-L1 adipocytes versus data demonstrating that Curcumin activates proliferation of 3T3-L1 adipocytes. In addition, the present studies aim to elucidate the underlying molecular mechanism utilized by Curcumin. The results showed that increasing doses of curcumin significantly inhibited 3T3L1adipocyte viability%. A lot of compelling evidences have also shown that curcumin exerts an angio-inhibitory action by decreasing vascular endothelial growth factor levels of NIH3T3 cells (17). These results were in agreement with previous finding that curcumin inhibited differentiation and increased apoptosis in 3T3-L1 (16) and in T -cell lymphoma cell lines (20). Curcumin can significantly decrease leptin release in LPS-treated adipocytes in concentration and time-dependent way (5). The action of Curcumin observed in our current study is in agreement to results published by Guo and coworkers (24) who treated human colorectal carcinoma cells with 0–30 µg/mL curcumin and observed significant concentration-dependent inhibition of cell viability and growth, as well as induction of apoptosis. It is worth mentioning that low doses of curcumin (0.02 µM for 24 h) exert different actions than high doses, which can have cytotoxic effects on 3T3-L1 cells (22).
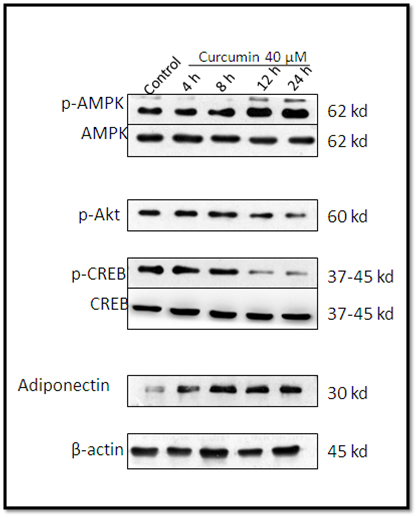
Figure 4.- Western blot analysis showed that curcumin (40 mM) treated 3T3L1 cells at the indicated time induced AMPK phosphorylation (p-AMPK) and inhibited phosphorylated CREB (p-CREB) and also phosphorylated AKt (p-Akt) to a lesser extent . Adiponectin expression increased with time increasing in compartion with control. The expression of AMPK and CREB were still detected and didn’t differ post curcumin (40µM) treatment in indicated time, β – actin used as control for protein loading
Furthermore, flow cytometry analysis of cells treated with curcumin revealed an accumulation of apoptotic cells and suggested a clear and gradual increase in cell death compared with control (Figure 3). The results were in agreement with those published by Kim et al (38); We hypothesize that this delay in cell growth may be attributable to re-regulation of the expression of some essential proteins required in cell cycle (39) and to the modulation in apoptotic factors (24). Curcumin has shown apoptotic effects in many cell lines in a concentration- and time-dependent manner (6).

Figure 5a.- Curcumin (40 µM) treatment showed clear decrease in the expression of PKA catalytic subunit with time increasing in 3T3L1 cells compared with control.
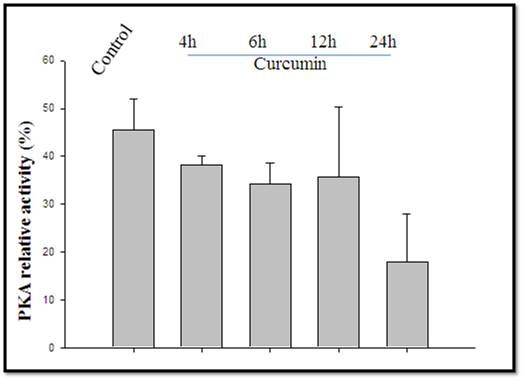
Figure 5b.- Curcumin (40 µM) treatment showed an inhibiting effect on PKA activity (%) during 4-24h on 3T3L1 cells.
In this study, FACS analysis showed that curcumin arrested the cell cycle. Curcumin was shown to induce sequential G1/S and G2/M phase arrest in a human osteosarcoma cell line (37) and S phase arrest in human colorectal carcinoma cells (24).Our data demonstrated found that Curcumin induced apoptosis and inhibited ROS generation level induced by the oxidant H2O2 in 3T3L1 adipocytes. This result could be explained by the findings of Balasubramanyam et al (40) who showed that the antioxidant effects of curcumin occurred via inhibition of Ca2+ influx and protein kinase C. In contrast, Thayyullathil et al (41) reported that curcumin induced ROS generation, which in turn resulted in apoptosis via activating apoptotic-inducing factor in mouse fibroblast L929 cells. In addition, Yang et al, (42) showed that ajoene exerts apoptotic activity in 3T3L1 cells by increasing intracellular ROS levels. The results of the current study demonstrate that curcumin exerts apoptotic effect on 3T3L1 cells in an ROS generation-independent fashion.
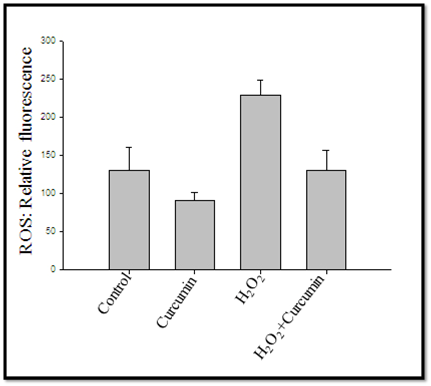
Figure 6.- Showed curcumin (40µM) inhibiting effect on ROS generation level induced with/ without H2O2 in 3T3L1 adipocytes.
Western blot analysis revealed that curcumin inhibited 3T3L1 viability and affected cellular signalling, including PKA pathway inhibitions, cyclic adenosine mono phosphate (cAMP) is an essential intracellular second messenger for controlling a variety of cellular processes, and PKA is its primary target (43). In this study, 40 µM curcumin increased p-AMPK levels (Figure 4) but inhibited expression of the PKA catalytic subunit and its relative activity (Figure 5). PKA has four subunits, and the dissociated active catalytic subunits elicit cellular responses by increasing the phosphorylation of several proteins. cAMP-mediated transcriptional responses requires the ability of PKA to phosphorylate CREB (30) . Induction of CREB is followed by the expression of many transcription factors that support adipogenesis (13,44,45). To identify the physiological importance of PKA signalling on the apoptotic action of curcumin, we examined the expression of PKA catalytic subunits in Curcumin treated 3T3L1 adipocyte cells. We demonstrated that Curcumin treatment blocked PKA catalytic subunit expression and inhibited PKA relative activity (Figure 5A), especially at 24h post treatment (Figure 5B),, which explained the gradual reduction in p-CREB levels (Figure 4). As Akt's phosphorylation is required for its efficient activation (30), the p-Akt expression was also examined and found that it has decreased but to a lesser extent than p-CREB (Figure 4). This means that the apoptotic action of curcumin on 3T3L1 cells is accompanied by decreasing p-CREB and p-Akt expression. This is consisting with the function of CREB in mediating Akt’s action described by Wang et al.(46) and Akt’s ability to phosphorylate CREB (47).
In contrast, these results disagree with the finding of Yeo et al (30) studying prostaglandin-mediated anti-apoptotic action on monocytic HL-60 cells.
Adiponectin hormone concentrations are inversely correlated with body fat % in adults (48). In transgenic mice, there was a reduction in adipocytes differentiation following overexpression of adiponectin targeted to adipose tissue (49) which is in agreement with other findings. Curcumin’s apoptotic effect was accompanied by increased expression of both adiponectin and p-AMPK (Figure 4). Adiponectin appears to be a growth antagonistic hormone in 3T3L1 adipocytes and may promote apoptosis via the AMPK signal pathway. AMPK has a key role in regulating cell metabolism, energy (50), cell growth, autophagy, and cell reprogramming, (12). In the current study, AMPK was detected in 3T3L1 before and 24-h post-curcumin treatment, a result that is supported by a previous study of adipocytes and cancer cells, which found that curcumin plays a critical role on cell differentiation and growth inhibition via AMPK activation (19, 12). Upregulation of fatty acid oxidation is a physiological function of leptin and adiponectin in skeletal muscle, and adipokines are able to carry out this function through their effect on AMPK signalling (51, 52). Our findings demonstrated that both adipokines and cAMP play a role in apoptosis and are supported by the study on breast cancer cells by Naviglio et al, (9) who concluded that although leptin inhibits adenylate cyclase/cAMP/PKA, elevating cAMP levels may be a novel therapy for breast cancer. Furthermore, Hardie et al (53) demonstrated that stimulators of AMPK may be of useful tools for preventing metabolic diseases.
In conclusion, curcumin modulates the phosphorylation of cellular signalling molecules. Curcumin’s ability to inhibit the phosphorylation of transcription factor in 3T3L1 could be attributed to the ability of AMPK to adapt to acute changes in the metabolic enzymes via direct phosphorylation and through decreased transcriptional response (54). In addition, AMPK reduced the expression of CREB targets (12), and Curcumin decreased the expression of the PKA catalytic subunit and inhibited levels of the phosphorylated protein factors p-CREB and p-Akt, Finally, the apoptotic effect of curcumin was synchronised with decreased expression of p-Akt, which is an important factor in cell survival and proliferation (47).
5. Acknowledgement
The author thanks Dr Adeeb Shehzad for his technical assistance and help with figures preparation, and gratefully acknowledges the efforts by Prof. Alexzander Asea and Dr. Badaruddin Abbasi for carefully proofreading the manuscript.
6. references
1. Elmore S. Apoptosis: A Review of Programmed Cell Death. Toxicologic Pathology 35, 495–516 (2007).
2. Tilg, H.; Moschen, AR. Adipocytokines, mediators linking adipose tissue, inflammation and immunity. Nature Reviews Immunology. 6, 772-783 (2006).
3. Aoki, N.; Jin-no, S.; Nakagawa, Y.; Asai, N.; Arakawa, E.; Tamura, N.; Tamura, T.; Matsuda, T. Identification and characterization of microvesicles secreted by 3T3-L1 adipocytes, redox- and hormone-dependent induction of milk fat globule-epidermal growth factor 8-associated microvesicles. JEndocrinology 148(8), 3850-3862 (2007).
4. Zeigerer, A.; Rodeheffer, M S.; McGraw, T E.; Friedman, J M. Insulin regulates leptin secretion from 3T3-L1 adipocytes by a PI 3 kinase independent mechanism. JExp Cell Res. 1, 314(11-12), 2249-2256 (2008).
5. Ciardi, C.; Jenny, M.; Tschoner, A.; Ueberall, F.; Patsch, J.; Pedrini, M.; Ebenbichler, C.; Fuchs, D. Food additives such as sodium sulphite, sodium benzoate and curcumin inhibit leptin release in lipopolysaccharide-treated murine adipocytes in vitro. Br J Nutr 107(6), 826-833 (2012).
6. Jiang, M C.; Yang-Yen, H F.; Yen, J J.; Lin, J K. Curcumin induces apoptosis in immortalized NIH 3T3 and malignant cancer cell lines. Nutr Cancer 26(1), 111-120 (1996).
7. Peyot, M.; Gadeau, A.; Dandré, F.; Belloc, I.; Dupuch, F.; Desgranges, C. Extracellular Adenosine Induces Apoptosis of Human Arterial Smooth Muscle Cells via A2b-Purinoceptor. Circulation Research 86, 76-85 (2000).
8. Zhang, J.; Wang, Q.; Zhu, N.; Yu, M.; Shen, B.; Xiang, J.; Lin A. Cyclic AMP inhibits JNK activation by CREB-mediated induction of c-FLIP(L) and MKP-1, thereby antagonizing UV-induced apoptosis. Cell Death Differ 15(10), 1654-1662 (2008).
9. Naviglio, S.; Di Gesto, D.; Romano, M.; Sorrentino, A.; Illiano, F.; Sorvillo, L.; Abbruzzese, A.; Marra, M.; Caraglia, M.; Chiosi, E.; Spina, A.; Illiano, G. Leptin enhances growth inhibition by cAMP elevating agents through apoptosis of MDA-MB-231 breast cancer cells. Cancer Biology & Therapy 8(12), 1183-1190 (2009).
10. Ruderman, N B.; Park, H.; Kaushik, V K.; Dean, D S.; Constant, S.; Prentki, M.; Saha, A K. AMPK as a metabolic switch in rat muscle, liver and adipose tissue after exercise. Acta Physiol Scand 178, 435–442 (2003).
11. Ruderman, N. B.; Saha, A. K.; Vavvas, D.; Witters, L. A. Malonyl-CoA, fuel sensing and insulin resistance. Am J Physiol 276, E1–18 (1999).
12. Mihaylova, M. M.; Shaw, R. J. The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nature Cell Biology 13, 1016-1023 (2011).
13. Reusch, J. B.; Colton, L. A.; Klemm, D. J. CREB Activation induces adipogenesis in 3T3-L1 cells. Mol Cell Biol 20.3, 1008–1020 (2000).
14. Simon, H. U.; Haj-Yehia, A.; Levi-Schaffer, F. Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 5.5, 415-418 (2000).
15. Antognelli, C.; Gambelunghe, A.; Talesa, V. N.; Muzi, G. Reactive oxygen species induce apoptosis in bronchial epithelial BEAS-2B cells by inhibiting the antiglycation glyoxalase I defence, involvement of superoxide anion.; hydrogen peroxide and NF-κB. Apoptosis (2013 September 4) (Epub ahead of print).
16. Ejaz, A.; Dayong, W.; Paul, K.; Mohsen, M. Curcumn inhibits adipogenesis in 3T3-L1 adipocytes and angiogenesis and besity in C57/BL Mice1–3. The Journal of Nutrition 139 919–925 (2009).
17. Gururaj, A. E.; Belakavadi, M.; Venkatesh, D. A.; Marmé, D.; Salimath, B. P. Molecular mechanisms of anti-angiogenic effect of curcumin. Biochem Biophys Res Commun 4, 297(4), 934-942 (2002).
18. Kawamori, T.; Lubet, R.; Steele, V. E.; Kelloff, G. J.; Kaskey, R. B.; Rao, C. V.; Reddy, B. S. Chemopreventive effect of curcumin, a naturally occurring anti-inflammatory agent, during the promotion/progression stages of colon cancer. Cancer Res 59, 597–601 (1999).
19. Lee, YK.; Lee, W.S.; Hwang, J.T.; Kwon, D.Y.; Surh, Y.J.; Park, O.J. Curcumin exerts antidifferentiation effect through AMPKalpha-PPAR-gamma in 3T3-L1 adipocytes and antiproliferatory effect through AMPKalpha-COX-2 in cancer cells. J Agric Food Chem 14, 57(1), 305-310 (2009a).
20. Zhang, C.; Li, B.; Zhang, X.; Hazarika, P.; Aggarwal, B.B.; Duvic, M. Curcumin selectively induces apoptosis in cutaneous T-cell lymphoma cell lines and patients' PBMCs, potential role for STAT-3 and NF-kappaB signaling. J Invest Dermatol 130(8), 2110-2119 (2010)..
21. Masuelli, L.; Benvenuto, M.; Fantini, M.; Marzocchella, L.; Sacchetti, P.; et al. Curcumin induces apoptosis in breast cancer cell lines and delays the growth of mammary tumors in neu transgenic mice. J Biol Regul Homeost Agents 27(1), 105-119 (2013).
22. Kim, J H.; Park, S. H.; Nam, S. W.; Kwon, H. J.; Kim, B. W.; Kim, W. J.; Choi, Y. H. Curcumin stimulates proliferation, stemness acting signals and migration of 3T3-L1 preadipocytes. International Journal of Molecular Medicine 28(3), 429-435 (2011a).
23. Staal, S P.; Hartley, J. W.; Rowe, W. P. Isolation of transforming murine leukemia viruses from mice with a high incidence of spontaneous lymphoma. Proc. Natl. Acad. Sci 74(7), 3065–3067 (1977).
24. Guo, L D.; Chen, X. J.; Hu, Y. H.; Yu, Z. J.; Wang, D.; Liu, J. Z. Curcumin inhibits proliferation and induces apoptosis of human colorectal cancer cells by activating the mitochondria apoptotic pathway. Phytother Res 27(3), 422-430 (2013).
25. Ammon, H. P.; Wahl, M. A. Pharmacology of Curcuma longa. Planta Med 57, 1–7 (1991).
26. Chainani-Wu, N. Safety and Anti-Inflammatory Activity of Curcumin, A Component of Tumeric (Curcuma longa). The Journal of Alternative and Complementary Medicine 9.1, 161-168 (2003).
27. Nawaz, A.; Khan, G.; Hussain, A.; Ahmad, A.; Khan, A.; Safdar, M. Gomal. University Journal of Research 27(1), 7-14 (2011).
28. Lu, F.; Chen, H.; Zhou, C.; Liu, S.; Guo, M.; Chen, P.; Zhuang, H.; Xie, D.; Wu, S. T-type Ca2+ channel expression in human esophageal carcinomas, A functional role in proliferation. Cell Calcium 43(1), 49–58 (2008).
29. Lee, S.; Shehzad, A.; Jung, J.; Sonn, J.; Lee, J.; Park, J.; Lee, S. Protein kinase Cα protects against multidrug resistance in human colon cancer cells. Molecules and Cells 34(1), 61-69 (2012).
30. Yeo, H.; Shehzad, A.; Lee, Y. Prostaglandin E2 Blocks Menadione-Induced Apoptosis through the Ras/Raf/Erk Signaling Pathway in Promonocytic Leukemia Cell Lines. Molecules and Cells 33, 371-378 (2012).
31. Hsu; Yen. Posted without published information on the link http,//www.nchu.edu.tw/~add/budget/student%20abroad/inter-meeting-94/T94-3-27.pdf
32. Hsu, C.; Yen, G. Induction of cell apoptosis in 3T3-L1 pre-adipocytes by flavonoids is associated with their antioxidant activity. Molecular Nutrition & Food Research 50(11), 1072–1079 (2006).
33. Reyes-Leon, A.; Atherton, J C.; Argent, R H. Puente, J. L.; Torres V. Heterogeneity in the Activity of Mexican Helicobacter pylori Strains in Gastric Epithelial Cells and Its Association with Diversity in the cagA Gene. Infect. Immun 75(7), 3445-3454 (2007).
34. Blum, S.; Moore, A N.; Adams, F.; Dash, P K. A mitogen-activated protein kinase cascade in the CA1/CA2 subfield of the dorsal hippocampus is essential for long-term spatial memory. J. Neurosci 19, 3535-3544 (1999).
35. Roy, A.; Ganguly, A.; Dasgupta, S. B.; Das, B. B.; Pal, C.; Jaisankar, P.; Majumder H. K. Mitochondria-Dependent Reactive Oxygen Species-Mediated Programmed Cell Death Induced by 3.;3_-Diindolylmethanethrough Inhibition of F0F1-ATP Synthase in Unicellular Protozoan Parasite Leishmania donovani. Molecular Pharmacology 74(5), 1292-1307 (2008).
36. Wang, D.; Jiang, H. U.; Lin, L. V.; Xiuwen, X.I.A..; Jianzhong, L.I.U.; Xiaoyuan, L.I. Enhanced inhibitory effect of curcumin via reactive oxygen species generation in human nasopharyngeal carcinoma cells following purple-light irradiation. Oncology Letters 6(1), 81–85 (2013).
37. Lee, D. S.; Lee, M. K.; Kim, J. H. Curcumin induces cell cycle arrest and apoptosis in human osteosarcoma (HOS) cells. Anticancer research 29, 5039-5044 (2009b).
38. Kim, CY.; Le, T. T.; Cheng, J.; Kim, K. Curcumin inhibits adipocyte differentiation through modulation of mitotic clonal expansion. The Journal of Nutritional Biochemistry 22(10), 910-920 (2011b).
39. Bertoli, C.; Skotheim, J. M.; De Bruin, R.A.M. Control of cell cycle transcription during G1 and S phases Nature Reviews. Molecular Cell Biology 14, 518- 528 (2013).
40. Balasubramanyam, M.; Koteswari, A. A.; Kumar, R. S.; Monickaraj, S. F.; Maheswari, J U.; Mohan, V. Curcumin-induced inhibition of cellular reactive oxygen species generation, Novel therapeutic implications. J. Biosci 28, 715–721 (2003).
41. Thayyullathil, F.; Shahanas, C.; Abdulkader, H.; Mahendra, P.; Sehamuddin, G. Rapid reactive oxygen species (ROS) generation induced by curcumin leads to caspase-dependent and -independent apoptosis in L929 cells. Free Radical Biology and Medicine 45(10), 1403–1412 (2008).
42. Yang, J.; Della-Fera, M. A.; Nelson-Dooley, C.; Baile, C. A. Molecular Mechanisms of Apoptosis Induced by Ajoene in 3T3-L1 Adipocytes. Obesity 14(3), 388–397 (2006).
43. Funamoto, S.; Anjard, C.; Nellen, W.; Ochiai, H. cAMP-dependent protein kinase regulates Polysphondylium pallidum development. Differentiation 71(1), 51-61 (2003).
44. Voet, V.; Pratt. Fundamentals of Biochemistry. Wiley; 2006.
45. Peverelli,E.; Ermetici, F.; Corbetta, S.; Gozzini, E.; Avagliano, L.; Zappa, M.; Bulfamante G.; Beck-Peccoz, P.; Spada, A.; Mantovani G. PKA regulatory subunit R2B is required for murine and human adipocyte differentiation. Endocr Connec EC-13-0049, (2006).
46. Wang, J. M; Chao, J. R.; Chen, W.; Kuo, M L.; Yen, J J.; Yang-Yen, H F. The antiapoptotic gene mcl-1 is up-regulated by the phosphatidylinositol 3-kinase/Akt signaling pathway through a transcription factor complex containing CREB. Mol. Cell Bio 19, 6195-7206 (1999).
47. Song, G.; Ouyang, G.; Bao, S. The activation of Akt/PKB signaling pathway and cell survival. Journal of Cellular and Molecular Medicine 9(1), 59–71 (2005).
48. Ukkola, O.; Santaniemi, M. Adiponectin: a link between excess adiposity and associated comorbidities? J. Mol. Med 80(11), 696–702 (2002)..
49. Bauche, I B.; El Mkadem, S A.; Pottier, A. M.; Senou, M.; Many, M. C.; Rezsohazy, R.; Penicaud, L.; Maeda, N.; Funahashi, T.; Brichard, S M. Overexpression of adiponectin targeted to adipose tissue in transgenic mice: impaired adipocyte differentiation. Endocrinology 148, 1539–1549 (2007).
50. Zhang, B. B.; Zhou, G.; Li, C. AMPK, an emerging drug target for diabetes and the metabolic syndrome. Cell Metab 5, 407-416 (2009).
51. Minokoshi, Y.; Kim, Y. B.; Peroni, O. D.; Fryer, L. G.; Müller, C.; Carling. D.; Kahn, BB. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature 415 (6869), 339–343 (2002).
52. Taylor, E B.; Hurst, D.; Greenwood, L J.; Lamb, J D.; Cline, T D.; Sudweeks, S N.; Winder, W W. Endurance training increases LKB1 and MO25 protein but not AMP-activated protein kinase kinase activity in skeletal muscle. Am. J. Physiol. Endocrinol. Metab 287(6), E1082- E1089 (2004).
53. Hardie, D. G.; Ross, F A.; Hawley, S A. AMP-activated protein kinase, a target for drugs both ancient and modern. Chem Biol 26,19(10), 1222-1236 (2012).
54. Cantó, C.; Auwerx, J. AMP-activated protein kinase and its downstream transcriptional pathways. Cell Mol Life Sci 67(20), 3407-3423 (2010).